Element cl neutrons1/20/2024  Now, you must be thinking about why one is added here well, it is because it has one negative charge that means the electron gain.Īfter calculating the protons, electrons, and neutrons, we have finally come to conclusive values of all. Now that we know the number of protons in Chlorine = 17Īs it was said earlier, if Chlorine has 17 protons, it needs to have the same number of electrons. Therefore, the number of neutrons in Chlorine= 18 Number of neutrons in Chlorine = Atomic mass number of Chlorine – Number of Protons Therefore to the number of neutrons, we can subtract the number of protons from the atomic mass number. A nucleus is a primary contributor to the atom’s mass which means that the mass number can identify the number of neutrons and protons present in an atom.Įach neutron and proton contains a relative mass of 1 unit. This indicates that each chlorine atom contains 17 protons and must also have 17 electrons.Īn atom’s nucleus contains neutrons and protons. Therefore if we take the case of Chlorine,Ĭalculating the number of neutrons, protons, and electrons in a Chlorine atom So, therefore, there must be an equal number of electrons and protons in an atom so that the charges cancel out. 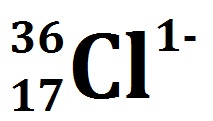
These weigh 0 amu.Įssential about protons, electrons, and neutrons ParticleĪs it is known that Protons carry +1 charge Its mass is equal to the mass of a proton and weighs one amu.Įlectrons are negatively charged particles surrounded by the nucleus. Neutrons are the no charged subatomic particles forming the part of the nucleus of an atom. It also determines the atomic number of an element and weighs one amu. Protons are positively charged subatomic particles that form the part of a nucleus of an atom. 
Before moving further, let us get acquainted with the definition of all three terms. However, the charge on both electrons and protons is the same but opposite. Study with Quizlet and memorize flashcards containing terms like The smallest unit which retains the properties of an element is a (n), Atomic mass is determined by the number of protons plus, Complete. Choose the total number of each item requested below. The neutrons and protons are present in the center of the atom that makes up the nucleus.Ī proton contains a positive charge, whereas electrons are negatively charged. Naturally occurring compounds in the earth are called. 
Atoms are generally made of extremely tiny particles known as neutrons, electrons, and protons. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
